A 4-year-old boy of Bangladeshi origin was referred to paediatric endocrine clinic by his community paediatrician due to concerns of precocious puberty.
Read the patient information and answer the questions below.
- Mother noticed pubic hair development and penis enlargement from 3.5 years of age.
- She had also noticed that he had begun to develop some sexual seeking behaviour and had become more disruptive, aggressive and angry in his behaviour.
- She had not noticed any underarm hair.
- No history of headaches or any visual problems or neurological symptoms.
- No history of abdominal pain.
- He was born in good condition at 39 weeks’ gestation via emergency Caesarean section for failure to progress, weighing 3.15 kg. Normal newborn check.
- At 6 weeks of age, he was brought to A&E via ambulance due to a history of fever and becoming unresponsive. He presented with tachycardia and hypoxia and was treated for confirmed streptococcus gallolyticus septicaemia (blood culture positive, cerebrospinal fluid culture negative). He responded well to antibiotics with good recovery.
- At 16 months of age, he was brought to A&E with a history of fever and vomiting and treated for E. coli urinary tract infection with complete resolution. Ultrasound scan of his kidneys at the time was normal.
- He was under the community paediatricians for moderate global developmental delay and severe speech and language delay. His referral was accompanied by a neurodevelopmental assessment which reported that he communicated in mostly single words and echoed short phrases. He had started to speak in short two-word phrases, for example “get up”. He displayed good eye contact and social smiling. He was independently mobile and could walk up and down stairs but not yet walk on tip toes. He could throw and catch a ball two-handed and kick a ball. He was able to draw a horizontal and vertical lines and circular scribbles but not yet imitate a circle.
- Two-year-old brother who is well and developing normally.
- Non consanguineous family.
- No family history of precocious or delayed puberty or other endocrine disorders.
- Auxology:
- Weight 28.3 kg (+3.63 SDS)
- Height 127 cm (+5.59 SDS)
- BMI 17.55 kg/m² (+1.25 SDS)
- Cardiovascular, respiratory, abdominal and neurological examinations: normal.
- Notable speech delay.
- No hyperpigmented patches or peripheral stigmata of chronic disease.
- Pubertal status: Gonads 3 Pubic hair 3 Axillary hair 1 with right testicular volume of 3 mL and a left testicular volume of 2 mL.
Question 1.
What are your differential diagnoses?
- Central precocious puberty (GRH-dependent)
- Pseudo precocious puberty (GnRH-independent)
- Simple virilising congenital adrenal hyperplasia
- Adrenal tumour
- McCune Albright Syndrome
- Obesity
- Hyperthyroidism
- GH excess
- Kleinfelter syndrome
- Simple virilising congenital adrenal hyperplasia
- Adrenal tumour
Question 2.
What investigations are required to tailor your diagnosis?
- Full blood count (FBC), Renal function tests/urea and electrolytes (U&Es), Liver function tests (LFTs)
- Luteinising hormone (LH), Follicular stimulating hormone (FSH), testosterone
- Adrenocorticotropic hormone (ACTH), cortisol, aldosterone, androstendione, 17OH-Progesterone, dehydroepiandrosterone sulphate (DHEAS)
- Thyroid stimulating hormone (TSH), free thyroxine (FT4)
- Insulin like growth factor-1 (IGF-1)
- Ultrasound abdomen
- Bone age
- Full blood count (FBC), Renal function tests/urea and electrolytes (U&Es), Liver function tests (LFTs)
- Luteinising hormone (LH), Follicular stimulating hormone (FSH), testosterone
- Adrenocorticotropic hormone (ACTH), cortisol, aldosterone, androstendione, 17OH-Progesterone, dehydroepiandrosterone sulphate (DHEAS)
- Thyroid stimulating hormone (TSH), free thyroxine (FT4)
- Insulin like growth factor-1 (IGF-1)
- Ultrasound abdomen
- Bone age
- FBC, U&Es, LFTs normal
- Puberty hormones:
- FSH <1 unit/L (1.5–12.4)
- LH <1 unit/L (1.7–8.6)
- Testosterone <0.5 nmol/L
- Normal values by Tanner staging:
- 1: <0.5
- 2: <0.5–15
- 3: 3.3–27
- Normal values by Tanner staging:
- Androgen panel
- ACTH 76 ng/L (high; normal at 09:00 <50 ng/L)
- Cortisol 194 nmol/L (lab comments: cortisol >550 nmol/L makes adrenal insufficiency unlikely)
- Aldosterone 1422 pmol/L (normal range: 150–550 supine, 250–950 upright)
- Androstenedione 26.6 nmol/L
- Pre-pubertal (0.5–9y): <0.8 nmol/L
- Post pubertal: 2–5.4 nmol/L
- 17OHP 162 nmol/L (follicular & adult males 1–8.7 nmol/L)
- DHEAS 5.8 nmol/L
- Pre-pubertal range (0.5–9 years): < 0.5 µmol/L
- Post-pubertal range: Male 2.3–10.0 µmol/L, Female 1.6–7.8 µmol/L
- T4 15.8, TSH 2.65 (normal)
- IGF-1 380 µg/L (normal)
- US abdomen: prominent adrenal glands, no adrenal masses
- Bone age: The patient’s chronological age at the time of the radiograph is 4 years and 4 months. Bone age is 13 years and 6 months (two standard deviations is 14 months). Conclusion: Very advanced bone age.
Question 3.
What additional investigations would confirm your diagnosis?
- Short synacthen test including 17OH-P levels
- Urinary steroid profile
- Genetics for CYP21 mutation
- MRI brain
- Short synacthen test including 17OH-P levels
- Urinary steroid profile
- Genetics for CYP21 mutation
- Short Synacthen test:
- Cortisol levels (nmol/L)
- 0 min: 209
- 30 min: 245
- 60 min: 247
- 17OHP levels:
- 0 min: 445
- 30 min: 591
- 60 min: 548
- Interpretation: elevated baseline 17OHP and response to short synacthen test
- Cortisol levels (nmol/L)
- Urinary steroid profile: Excess 17-hydroxyprogesterone metabolites. DHA, DHA metabolites and pregnenalone are at relatively high concentrations, as are the androstenedione metabolites. All would be a consequence of prolonged adrenal hyperstimulation.
- Genetics report: Heterozygous for CYP21A2 c.290-13C>G and c.515T>A mutations
Question 4.
What is the diagnosis?
- Central precocious puberty
- CAH presenting with GnRH-independent precocious puberty
- Adrenal tumour
- CAH presenting with GnRH-independent precocious puberty
Treatment:
- Commenced on hydrocortisone 10 mg/m2 per day
- Re-assessed 3 months after starting hydrocortisone treatment:
- Height + 1.2cm
- Pubertal staging: P4 G3 A1 with 5 mL testes bilaterally
- Due to ongoing evidence of precocious puberty, he underwent LHRH testing:
Time (mins) 0 30 60 FSH 1.9 2.5 3.3 LH 2 13 14
Question 5.
What does this show?
- Normal response
- Central precocious puberty
- Gonadotrophic independent precocious puberty
- Central precocious puberty
Treatment:
- Hydrocortisone 5 mg/2.5 mg/2.5 mg = 10 mg/m2 per day
- GnRH analogue (Triptorelin 3 monthly)
Discussion
This patient presented with late virilising congenital adrenal hyperplasia (CAH). CAH is caused by defects in one of the enzymes involved in the steroidogenic pathway. The precise mode of presentation and clinical and biochemical features are determined by the nature and severity of the specific “block”. 95% of CAH cases are caused by 21 hydroxylase deficiency. The inability to convert 17‐OHP to 11‐deoxycortisol results in cortisol deficiency and shunting of precursors into androgen pathways (figure 1).
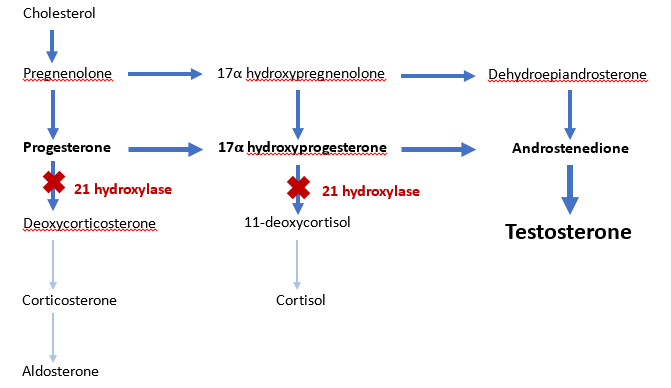
Figure 1: Deficiency of 21-hydroxylase leads to an accumulation of precursor steroids and shunting to the androgen pathway.
CAH is a recognised cause of precocious pseudopuberty and some children with CAH also develop true central precocious puberty with early maturation of the hypothalamic-pituitary-gonadal (H-P-G) axis. In this case, the patient presented with precocious pseudopuberty (evidenced by pre-pubertal testes volume of <4 mL) and was later found to have true central precocious puberty with enlargement of testicular volume. Progressive precocious puberty can have consequences for growth and psychosocial development. Accelerated bone age can lead to premature fusion of growth plates and short final height.
Key take home points:
- Definition of precocious puberty: <8 years old in girls and <9 years old in boys
- Always examine the genitalia in children presenting with tall stature.
- Careful pubertal staging and testicular size will determine if the cause of precocious puberty is central (gonadotropin-dependent), involving activation of the H-P-G axis or peripheral (gonadotropin-independent), involving autonomous activation of hormone production (either from the adrenal glands or gonads)
- CAH can present in childhood with pseudo/peripheral precocious puberty but patients can also develop central precocious puberty which requires additional treatment.
Reference
Hughes CR, Man E, Achermann JC. The Adrenal Cortex and Its Disorders. Brook’s Clinical Pediatric Endocrinology; 2019: 335–407


