Case study introduction
A 5‑year‑old girl is brought to clinic with a 2.5‑year history of intermittent headaches that have gradually increased in both frequency and intensity. Over the 3 weeks prior to presentation, her symptoms escalated significantly. She has experienced severe headaches accompanied by episodes of vomiting, after which the headache temporarily improves. Her parents report that she has become increasingly distressed, withdrawn, and noticeably low in mood during this period. They have also observed subtle cognitive changes, including difficulty recalling familiar information such as the names of her favourite toys. In the week before presentation, she began complaining that reading her books had become difficult because her vision appeared blurred. She had increased thirst and urinary frequency for several months earlier, which resolved spontaneously a few weeks before the current presentation. There is no history of recent trauma, fever, or seizures.
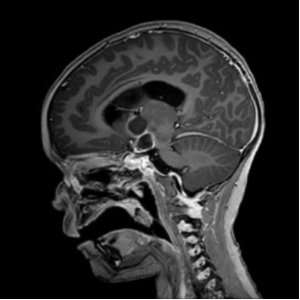 Figure 1 – MRI scan showing suprasellar craniopharyngioma. Reproduced with parental permission (2025)
Figure 1 – MRI scan showing suprasellar craniopharyngioma. Reproduced with parental permission (2025)
1.What does the MRI show?
The child’s presentation and MRI findings are highly consistent with a suprasellar craniopharyngioma, a locally aggressive tumour arising from remnants of Rathke’s pouch.
Explanation
- In this case, the tumour’s location and mass effect explain the full spectrum of her symptoms. Her long‑standing headaches, which have gradually worsened over more than 2 years, together with vomiting that temporarily relieves the pain, strongly suggest raised intracranial pressure due to obstruction of cerebrospinal fluid flow. This is typical of craniopharyngiomas that compress the third ventricle or block the foramen of Monro.
- Her recent blurred vision and difficulty reading are characteristic of optic chiasm compression, a common consequence of the tumour’s proximity to the visual pathways. These visual symptoms often emerge as the tumour enlarges or a cyst expands.
2. What further investigations would you like to perform?
1. Full endocrine evaluation
Craniopharyngiomas commonly affect the hypothalamic–pituitary axis. Given her history (thirst, urinary frequency, behavioural change, memory issues), a comprehensive endocrine work‑up is essential.
Baseline tests:
- Serum morning cortisol or standard dose synacthen test
- Adrenocorticotropic hormone (ACTH)
- Thyroid function tests (thyroid-stimulating hormone [TSH], free thyroxine [free T4])
- Growth hormone (GH) axis (IGF‑1, IGFBP‑3)
- Prolactin
- Gonadotropins (luteinising hormone [LH], follicle-stimulating hormone [FSH])
- Serum sodium, plasma and urine osmolality (to assess for arginine vasopressin [AVP] insufficiency)
- Consider water deprivation test only if safe and needed after initial lab results
- Routine blood tests (full blood count, urine and electrolytes, liver function tests, clotting) for pre-operative assessment
2. Formal ophthalmology assessment
Her blurred vision and difficulty reading strongly suggest visual pathway involvement.
Assessments:
- Visual acuity
- Visual fields (age‑appropriate methods)
- Fundoscopy for papilloedema or optic atrophy
Her blood tests showed the following:
Cortisol (>430 nmol) | GH (>7 μg/L) | ACTH (7.2–63.3 ng/L) | TSH (0.76–5.91 mIU/L) | T4 (11.6–21.6 pmol/L) | |
Baseline | 7 | 1.10 | 4.0 | 1.55 | 9.8 |
30 min | 60 | 1.20 | |||
60 min | 73 | 0.82 | |||
90 min | 0.99 | ||||
120 min | 0.75 | ||||
150 min | 0.75 |
Table 1. Blood test results for patient with craniopharyngioma. Expected range and units shown in brackets.
3. What do these blood test results suggest?
The results indicate anterior pituitary dysfunction, and when considered alongside her increased thirst and polyuria, they are consistent with panhypopituitarism.
4. Are you worried about her behavioural change?
- Yes, her behavioural change, withdrawal, and difficulty recalling familiar information may reflect hypothalamic involvement.
- Furthermore, increasing distress, low mood, and cognitive slowing further support the likelihood of hypothalamic compression, which can affect emotional regulation, appetite, sleep, and memory.
- These symptoms often evolve gradually but may accelerate with tumour growth.
5. The patient was administered hydrocortisone on admission. Shortly after, she began passing large volumes of urine more frequently. What is the most likely cause of her increased urine output?
- Osmotic diuresis due to hyperglycaemia
- Nephrogenic diabetes insipidus (DI)
- Unmasked central DI due to AVP deficiency
- Syndrome of inappropriate antidiuretic hormone secretion (SIADH)
Correct Answer: Unmasked central DI due to AVP deficiency
Explanation:
Craniopharyngiomas often affect the hypothalamic–pituitary axis, and AVP (antidiuretic hormone) deficiency is a common consequence. In stressed or acutely ill patients, endogenous cortisol deficiency can mask AVP insufficiency by reducing renal free water clearance. When hydrocortisone is administered, it restores normal renal responsiveness, thereby unmasking the underlying central diabetes insipidus. This leads to polyuria and polydipsia due to the lack of AVP-mediated water reabsorption in the kidneys. This phenomenon is well-recognised in patients with pituitary tumours.
POSTOPERATIVE PATTERS
The patient underwent surgical resection of a craniopharyngioma. Postoperatively, she initially developed high serum sodium and high urine output. Three days later, she presented with low serum sodium, low urine output, and high urine osmolality. Subsequently, she developed high urine output again, with low urine osmolality and high serum osmolality.
6. What is the most likely explanation for this sequence of events?
- SIADH
- Triphasic response following pituitary surgery
- Intermittent dehydration following surgery
- Cerebral salt wasting with intermittent AVP insufficiency
Correct Answer: Triphasic response following pituitary surgery
Explanation:
This pattern reflects the classic triphasic response seen after neurosurgical procedures involving the hypothalamic–pituitary region, especially craniopharyngioma resection[1]:
Phase 1: transient DI
Initial damage to AVP-producing neurons leads to transient central DI, causing polyuria and hypernatremia.
Phase 2: SIADH-like symptoms
SIADH-like phase: Damaged neurons release stored AVP uncontrollably, resulting in water retention, hyponatremia, low urine output, and concentrated urine.
Phase 3: permanent DI
If AVP-producing cells are irreversibly damaged, the patient enters chronic central DI, with polyuria, hypo-osmolar urine, and hypernatremia.
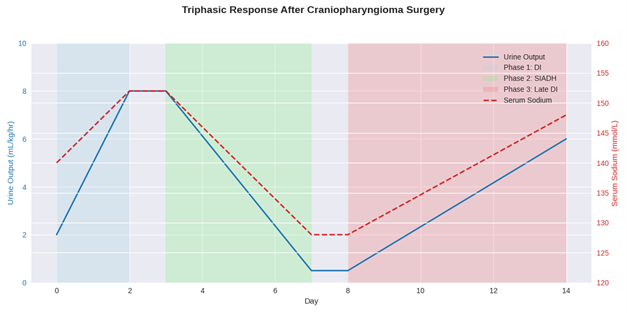
Figure 2 – The triphasic response after craniopharyngioma surgery
Recognising this triphasic pattern is critical for appropriate fluid and endocrine management postoperatively, particularly in the first week following surgery. Strict fluid input and urine output with regular monitoring, or serum sodium and osmolarity paired with urine osmolarity, is key to ensuring that these phases can be identified.
SUBSEQUENT MANAGEMENT
Following pituitary surgery, the patient was treated with full anterior and posterior pituitary hormone replacement.
7. What hormones would you chose to replace?
Correct Answer:
Full pituitary hormone replacement involves:
- hydrocortisone for ACTH deficiency
- levothyroxine for TSH deficiency
- sex steroids for LH/FSH deficiency
- recombinant GH for GH deficiency
- desmopressin replaces vasopressin in AVP insufficiency
This regimen restores endocrine function and requires careful monitoring to mimic physiological balance.
INCREASING WEIGHT GAIN
Despite full pituitary hormone replacement, the 6-year-old girl underwent significant weight gain in the subsequent 6 months, and a rapid and marked increase in her body mass index (BMI) as demonstrated on the BMI chart below [Figure 3]
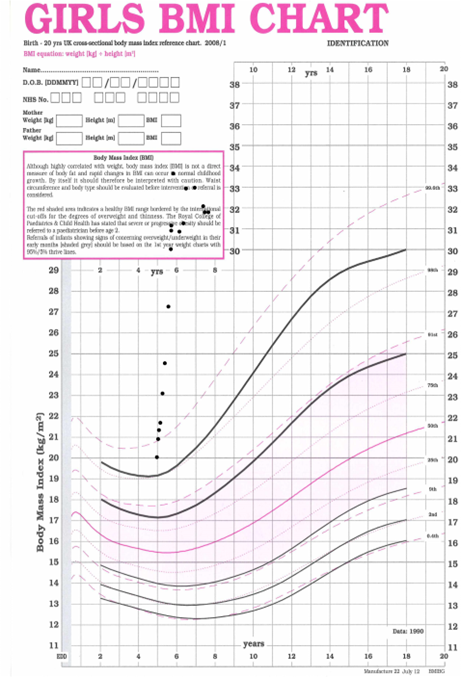
Figure 3. BMI growth chart for craniopharyngioma patient. Generated using Microsoft Copilot 2025.
8. Are you concerned about this weight gain?
Yes, significant weight gain in a 6-year-old following craniopharyngioma surgery and full pituitary hormone replacement is a clinical concern, particularly in the context of hypothalamic involvement [2, 3]. Craniopharyngiomas often affect the hypothalamus, which plays a central role in energy balance, appetite regulation, and metabolic control. Damage to this region can lead to hypothalamic obesity, a form of rapid, treatment-resistant weight gain driven by impaired satiety signalling, reduced energy expenditure, and autonomic dysregulation.
Unlike typical obesity, this condition is not easily reversed with lifestyle interventions alone. Even with optimal hormone replacement, hypothalamic dysfunction can persist, and early weight gain may signal the onset of this challenging complication. It warrants close monitoring, early intervention with dietetic and behavioural support, and consideration of pharmacological or specialist obesity management strategies.
Overall management strategies for craniopharyngioma
Management of craniopharyngioma in children requires a multidisciplinary, staged approach focused on tumour control, endocrine replacement, and long-term quality of life [4]:
Neurosurgical resection: Aim for maximal safe tumour removal while preserving hypothalamic and pituitary function. Complete resection may not be feasible due to proximity of critical structures.
Radiotherapy: Used postoperatively and for residual/recurrent disease. Techniques like proton beam therapy help minimise damage to surrounding tissue.
Endocrine replacement: Most children require lifelong hormone replacement for deficiencies in cortisol, thyroid hormones, sex steroids, GH, and vasopressin.
Hypothalamic obesity management: Early intervention with dietetic support, physical activity, behavioural therapy, and in some cases pharmacological agents, although most interventions have not proved successful.
Visual and neurological monitoring: Regular assessment of visual fields, acuity, and neurocognitive function due to tumour location and treatment effects.
Psychosocial support: Ongoing psychological care, educational support, and family counselling to address emotional, behavioural, and developmental challenges.
Transition to adult services: Structured transition planning ensures continuity of care into adulthood, especially for endocrine and neuropsychological follow-up.
- Finken MJJ, Zwaveling-Soonawala N, Walenkamp MJE, et al. Frequent Occurrence of the Triphasic Response (Diabetes Insipidus/Hyponatremia/Diabetes Insipidus) after Surgery for Craniopharyngioma in Childhood. Horm Res Paediatr 2011; 76(1): 22–26 https://doi.org/10.1159/000324115
- Dimitri P. The management of hypothalamic obesity in craniopharyngioma. Best Pract Res Clin Endocrinol Metab 2025; 39(5): 102018 https://doi.org/10.1016/j.beem.2025.102018
- Otte A, Müller HL. Childhood-onset Craniopharyngioma. J Clin Endocrinol Metab 2021; 106(10): e3820–e3836 https://doi.org/10.1210/clinem/dgab397
- Dimitri P. Treatment of Acquired Hypothalamic Obesity: Now and the Future. Front Endocrinol 2022; 13: 846880 https//doi.org:/10.3389/fendo.2022.846880


