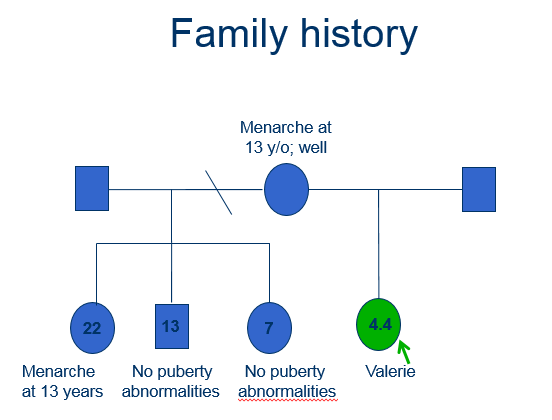
Case history
A 4-year-old girl – “Valerie” – is referred to the local paediatric endocrine unit with signs of pubertal development.
Female
Age 4.0 years of age
Presented to the Local Paediatric Endocrine clinic
Complaining of:
- Per vagina (PV) bleeding
- 1 episode
- Lasted for 3 days, requiring x 3 sanitary towels a day
- Minimal lower abdominal pain
- Breast development: progressed over the last year
- Axillary and pubic hair development: more recent
- Well looking
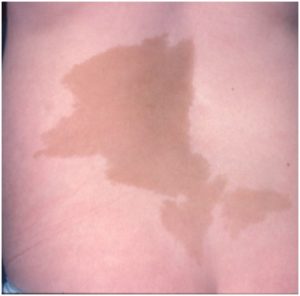
- No café-au-lait spots
- Abdomen slight distension
- suprapubic mass arising from the pelvis
- smooth, mobile, non-tender
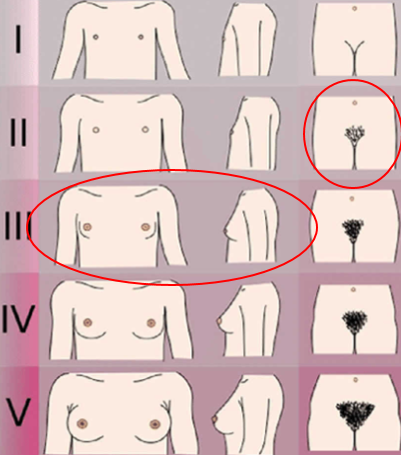
- Pubertal staging (Tanner)
- Breast stage 3 (B3)
- Axillary hair stage 2 (A2)
- Pubic hair stage 2 (P2)
- Vagina appeared oestrogenised
- All other systems unremarkable
Tanner stages of puberty
Question 1.
In girls, which endocrine organs control breast development and which control pubic hair development, respectively?
- Breast development controlled by adrenal glands and pubic hair growth controlled by ovaries
- Breast development controlled by parathyroid glands and pubic hair growth controlled by ovaries
- Breast development controlled by ovaries and pubic hair growth controlled by parathyroid glands
- Breast development controlled by ovaries and pubic hair growth controlled by adrenal glands
Breast development controlled by ovaries and pubic hair growth controlled by adrenal glands
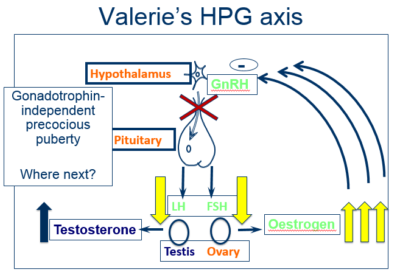
Different endocrine hormonal axes control the processes of breast development and pubic hair growth in girls.
- Breast development is controlled by oestrogen secretion from the ovaries under the control of the hypothalamic-pituitary-gonadal axis
- Growth of pubic and axillary hair is mainly under the influence of androgens secreted by the adrenal glands – so-called adrenarche
Question 2.
What is the significance of café-au-lait spots in assessment of a girl with signs of precocious puberty?
- That the early maturing girl is drinking too much coffee
- Along with precocious puberty, you will find café-au-lait spots in a rare condition called McCune-Albright syndrome
- Café-au-lait spots are a classic sign of precocious puberty caused by hyperthyroidism
- All the above
Along with precocious puberty, you will find café-au-lait spots in a rare condition called McCune-Albright syndrome
McCune-Albright syndrome is a genetic disorder that occurs in both boys and girls, which is characterised by three classic signs:
- Café-au-lait spots
- Bone disorder characterised by symptoms or signs on x-ray
- Endocrine gland hyperfunction; most commonly in girls this hyperfunction involves the ovary, resulting in gonadotrophin- independent precocious puberty
Anthropometrics
- Valerie
- Age: 4.0 years
- Height: 112.2 cm
- SD: + 2.6
- Mid-parental height target (MPHT) SD: –0.1
- tall for parents
- Weight: 22.8 kg
- BMI: 18.1
- BMI SD: 1.5
- Bone age x-ray: 6.5 years
- 2.5 years advanced
Question 3.
A 2.5 year advancement in bone age indicates which of the following is the most likely outcome:
- Valerie will stop growing earlier than her peers
- Valerie will stop growing at the same time as her peers
- Valerie will stop growing later than her peers
Valerie will stop growing earlier than her peers
- Bone age or skeletal maturity looks at how mature the bones are – or the degree of bone ossification – in comparison with the child’s chronological age
- The left hand and wrist is used, as a large number of bones can be x-rayed within a small area
- The two most common methods used to assessskeletal maturity are the atlas method (Greulich and Pyle) and the bone-specific scoring method (Tanner-Whitehouse)
Fusion of the growth plates is directly influenced by sex hormones, mainly oestrogens
Initial biochemistry
- Free (thyroxine (T4) 16.6 pmol/L (normal range: 10-24)
- Thyroid Stimulating Hormone (TSH): 3.2 mU/L (normal range: 0.2-5)
- Prolactin: 79 mu/L (normal range: <400)
- Testosterone: <0.7 nmol/L (normal range: <2.9)
- LH: <0.1 IU/L (normal pre-pubertal range: <1)
- FSH: <0.1 IU/L (normal pre-pubertal range: <1)
- Oestradiol 924 pmol/L (normal prepubertal range: 0-50)
GnRH stimulation test
GnRH stimulation test (2.5 µg/kg)
| Time (mins) | LH | FSH |
|---|---|---|
| 0 | < 0.1 | < 0.1 |
| 20 | < 0.1 | < 0.1 |
| 60 | < 0.1 | < 0.1 |
Question 4.
Looking at both the results of the GnRH stimulation test and the oestrogen level, what sort of response has Valerie had:
- A pre-pubertal response
- A pubertal response
- Oestrogen production not stimulated by the pituitary gland, ie, independent of the hypothalamic-pituitary-gonadal axis
Oestrogen production not stimulated by the pituitary gland, ie, independent of the hypothalamic-pituitary-gonadal axis
| Category | Response to GnRH (LHRH) |
|---|---|
| Normal individuals | FSH and LH values increase at 20 minutes and decrease again at 60 minutes |
| Gonadotrophin-dependent sexual precocity | Pubertal response |
| Gonadotrophin-independent sexual precocity | The levels of gonadotrophin are suppressed |
| Gonadal failure | The basal values are increased, and the response to GnRH is exaggerated |
| Pubertal delay | The basal values to GnRH is abssent or poor |
| Hypogonadotrophic | The basal values to GnRH is abssent or poor |
Valerie’s results reveal suppressed gonadotrophin levels, in conjunction with an elevated oestrogen level.
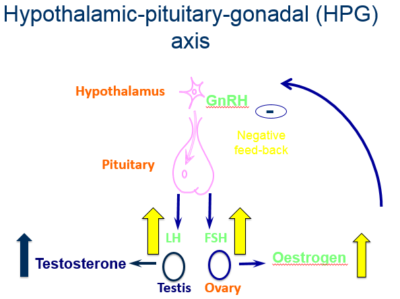
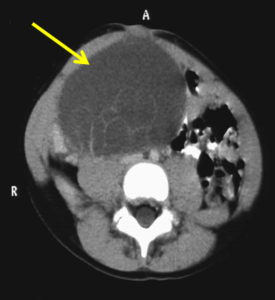
- Abdominal and pelvic Magnetic Resonance Imaging (MRI) scan
- Scan revealed a large mass approximately 15 cm x 10 cm
Summary
- Clinically
- 4.0 year old girl presenting with vaginal bleeding
- Tall stature: 112.2cm (SD: + 2.6)
- Tall for parents (mid-parental height target SD: -0.1)
- Pubertal staging (Tanner): B3, A2, P2
- Suprapubic mass arising from the pelvis
- Biochemistry
- Elevated oestrogen levels
- Negative LH, FSH response to GnRH stimulation
- Gonadotrophin-independent sexual precocity
- Imaging
Advanced bone age (2.5 years)
MRI: large pelvic mass of 15 cm x 10 cm
Question 5.
What is the best treatment option for Valerie:
- Refer to surgeons for assessment for potential surgical removal of mass
- Treat with a GnRH analogue that would suppress LH and FSH production from the pituitary
- Watch and wait to see if mass resolves
Refer to surgeons for assessment for potential surgical removal of mass
- Surgery is the treatment of choice for adrenal and ovarian tumours, or of large cysts which are so large that torsion or rupture may occur
- A GnRH analogue is NOT indicated as gonadotropins are already suppressed and not the cause of high oestrogen levels
Management
- Referral was made to the Paediatric Surgical team
- Surgery: laparotomy
- Right oophorectomy
- 14 x 10 cm cystic mass replacing right ovary
- Histology: cystic in nature, benign
- Left ovary normal
- 14 x 10 cm cystic mass replacing right ovary
- Post operatively: regression of pubertal symptoms and cessation of vaginal bleeding
- Clinic follow-up to ensure normal pubertal progression
- Sex steroids from the adrenal glands
- Congenital adrenal hyperplasia
- Adrenal tumour
- Premature adrenarche (< 6 years)
- Cushing’s syndrome
- Sex steroids from gonads
- Ovarian tumour, cysts
- McCune-Albright syndrome
- Testotoxicosis
- Human chorionic gonadotropin (hCG) secreting tumour
- Exposure to exogenous steroids
Conclusions
- Important to have a good understanding of normal pubertal progress and the physiology of the hypothalamic-pituitary-gonadal axis
- Diagnosis is achieved through a step-by-step process
- Importance of a good history, including family history of puberty
- Always consider the child / young person’s understanding, not just the parents
- Disorders of puberty may be complex!



